
Conductometric determination of sibutramine HCl, sumatriptan succinate and lomefloxacine HCl and the solubility products of their ion associates with molybdophosphoric acid
Main Article Content
Abstract
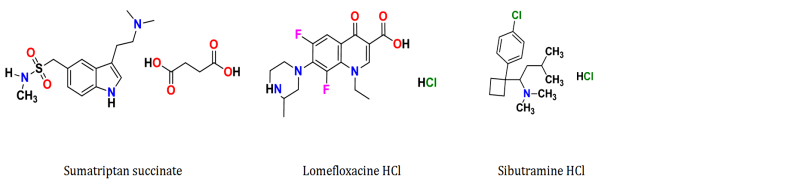
Conductometric determination of sibutramine HCl, sumatriptan succinate and lomefloxacine HCl with molybdophosphoric acid as a precipitating reagent was investigated. Various experimental conditions were evaluated and results obtained showed good recoveries, (mean recovery values of 100.51, 99.68, 99.41 and relative standard deviation of 0.332, 0.404, 0.509 for sibutramine HCl, sumatriptan succinate and lomefloxacine HCl, respectively). Numerical derivatization (first and second derivative) of the data was also applied, showing more accurate results compared to classical ones. The described procedures allowed the determination of equilibrium constants those indicated high degree of completeness of the precipitation reaction. Other parameters related to ion pair complex such as solubility and solubility product were also calculated. The described procedures allowed the determination of the studied drugs in the range of 5-15 mg. The precipitate obtained by ion pairing of lomefloxacine HCl with molybdophosphoric acid was spectroscopically characterized using IR. The method was further applied successively to pharmaceutical formulations, the proposed method offering a high degree of accuracy and precision when compared to reference methods.
Article Details
[1]. Sweetman, S. C. Martindale-The Complete Drug Reference, 36th edition, The Pharmaceutical Press, London, 2009.
[2]. Ariburnu, E.; Uludag, M.; Yalcinkaya, H.; Yesilada, E. J. Pharmaceut. Biomed. 2012, 64-65, 77-81.
http://dx.doi.org/10.1016/j.jpba.2012.02.004
PMid:22410500
[3]. Chorilli, M; Bonfilio, R.; Chicarelli, R. S.; Salgado, H. R. N. Anal. Method. 2011, 3, 985-990.
http://dx.doi.org/10.1039/c0ay00598c
[4]. El-Gohary, N. A.; El-Nashar, R. M.; Aboul-Enien, H. Y. Anal. Lett. 2011, 44, 241-257.
http://dx.doi.org/10.1080/00032719.2010.500760
[5]. Valarmathi, R.; Karpagam, K. S. S.; Revanthi, R. The Indian Pharm. 2004, 3, 71-72.
[6]. Maluf, D. F.; Farago, P. V.; Barreira, S. M. W.; Pedroso, C. F.; Pontarolo, R. Lat. Am. J. Pharm. 2007, 26, 909-912.
[7]. The United States Pharmacopiea 30, National Formulary 25, US Pharmacoppieal Convention, Rockville, MD, 2007.
[8]. Cheng, K. N.; Redrup, M. J.; Barrow, A.; Williams, P. N. J. Pharm. Biomed. Anal. 1998, 17, 399-408.
http://dx.doi.org/10.1016/S0731-7085(97)00220-3
[9]. Tipre, D. N.; Vavia, P. R. Indian Drugs. 1999, 36, 501-505.
[10]. Riddhi, G; Dharamsi, A. J. Drug Delivery Ther. 2013, 3, 93-97.
[11]. Sagar, K.; Alvarez, J. M. F.; Hua, C.; Smyth, M. R.; Munden, R. J. Pharmaceut. Biomed. 1992, 10, 17-21.
http://dx.doi.org/10.1016/0731-7085(92)80005-8
[12]. Ramu, B. K.; Raghubabu, K. Int. J. Appl. Biol. Pharm. Technol. 2011, 2, 86-91.
[13]. Ramu, B. K.; Raghubabu, K. Int. J. Pharm. Biomed. Sci. 2010, 1, 49-52.
[14]. Ramu, B. K.; Raghubabu, K. Int. J. Pharm. Pharm. Sci. 2011, 3, 175-178.
[15]. Ramu, B. K.; Raghubabu, K. Der Pharma Chemica 2011, 3, 223-228.
[16]. Satyanarayane, K. V. V.; Rao, P. N. E-J. Chem. 2011, 8, 269-275.
[17]. Ayad, M. M.; Abdellatef, H. E.; Hosny, M. M.; Kabil, N. A. Zag. J. Pharm. Sci. 2011, 20, 9-21.
[18]. Chitlang, S. S.; Ranjane, M.; Wankhede, S. B.; Sakarkar, D. M. Int. J. Pharm. Tech. 2009, 1, 844-851.
[19]. Sun, H.; Li, L.; Su, M. Chromatographia 2008, 67, 399-405.
http://dx.doi.org/10.1365/s10337-008-0518-5
[20]. Vilchez, J. L.; Araujo, L.; Prieto, A.; Navalon, A. J. Pharmaceut. Biomed. 2001, 26, 23-29.
http://dx.doi.org/10.1016/S0731-7085(01)00391-0
[21]. Limig, D.; Qingqin, X.; Jianmei, Y. Pharmaceut. Biomed. 2003, 33, 693-698.
http://dx.doi.org/10.1016/S0731-7085(03)00365-0
[22]. Du, L. M.; Yang, Y. Q.; Wang, Q. M. Anal. Chim. Acta 2004, 516, 237-243
http://dx.doi.org/10.1016/j.aca.2004.04.006
[23]. Geffken, D.; Salem, H. Am. J. Appl. Sci. 2006, 3, 1952-1960.
http://dx.doi.org/10.3844/ajassp.2006.1952.1960
[24]. Kaur, K.; Singh, B.; Malik, A. K. Thai J. Pharm. Sci. 2010, 34, 58-66.
[25]. Lu, J. Q.; Jin, F.; Sun, T. Q.; Zhou, X. W. Int. J. Biol. Macromol. 2007, 40, 299-304.
http://dx.doi.org/10.1016/j.ijbiomac.2006.08.010
PMid:17030362
[26]. Salem, H. Am. J. Appl. Sci. 2005, 2, 719-729.
http://dx.doi.org/10.3844/ajassp.2005.719.729
[27]. Gomes, G. C.; Salgado, H. R. N. Acta Farmaceutica Bon. 2005, 24, 406-408.
[28]. Askal, H.; Refaat, I.; Darwish, I.; Marzouq, M. Chem. Pharm. Bull. 2007, 55, 1551-1556.
http://dx.doi.org/10.1248/cpb.55.1551
[29]. Issa, Y. M.; Abdel-Gawad, F. M.; Abou-Table, M. A.; Hussein, H. M. Anal. Lett. 1997, 30, 2071-2084.
http://dx.doi.org/10.1080/00032719708001722
[30]. Amin, A. S.; Dessouki, H. A.; Agwa, I. A. Arabian J. Chem. 2008, 1, 209-215.
[31]. Suhagia, B. N.; Shah; S. A., Rathod, I. S., Patel, H. M.; Rao, Y. M. Indian J. Pharm. Sci. 2006, 68, 247-249.
http://dx.doi.org/10.4103/0250-474X.25728
[32]. Darwishm, I. A.; Sultan, M. A.; Al-Arfaj, H. A. Int. J. Res. Pharm. Sci. 2010, 1, 43-50
[33]. Elazazy, M. S.; Elmasry, M. S.; Hassan, W. S. Int. J. Electrochem. Sci. 2012, 7, 9781-9794.
[34]. Maria, S.; Dorneanu, V.; Ghimicescu, G. Talanta 1997, 24, 140-142.
[35]. Issa, Y. M.; Abdel-Ghani, N. T. Microchim. Acta 1999, 132, 83-88.
http://dx.doi.org/10.1007/PL00010077
[36]. Issa, Y. M.; Youssef, A. F. A.; Mutair, A. A. Il Farmaco 2005, 60, 541-546.
http://dx.doi.org/10.1016/j.farmac.2005.03.002
PMid:15890349
[37]. Lingane, J. J. Electroanalytical Chemistry, 2nd Edition, Interscience, New York, 1958.
[38]. Andropov, L. L. Theoretical Electrochemistry, Izdatelstvo Mir: Moscow, 1977.
[39]. Anis, S. M.; Hosny, M. M.; Abdellatef, H. E.; El- Balkiny, M. N. E-J. Chem. 2011, 8, 1784-1796.
[40]. Issa, Y. M.; Shoukry, A. F.; El-Nashar, R. M. J. Pharmaceut. Biomed. 2001, 26, 379-386.
http://dx.doi.org/10.1016/S0731-7085(01)00415-0
[41]. Ayad, M. M.; Abdellatef, H. E.; Hosny, M. M.; Kabil, N. Int. J. Pharm. Biomed. Res. 2012, 3, 121-126.
Department Faculty of Pharmacy, Zagazig University, Zagazig, Egypt
Most read articles by the same author(s)
- Magda Mohamed Ayad, Hisham Ezzat Abdellatef, Mervat Mohamed Hosny, Yassmin Ahmed Sharaf, Application of 4-chloro-7-nitrobenzofurazan for the analysis of propafenone and diltiazem hydrochlorides using kinetic spectrophotometric and spectrofluorimetric methods , European Journal of Chemistry: Vol. 4 No. 1 (2013): March 2013
- Magda Mohamed Ayad, Hisham Ezzat Abdellatef, Mervat Mohamed Hosny, Yassmin Ahmed Sharaf, Conductometric titration method for determination of naftidrofuryl oxalate, propafenone HCl and sotalol HCl using silver nitrate , European Journal of Chemistry: Vol. 3 No. 3 (2012): September 2012
Downloads
Metrics
License Terms
Copyright © 2026 by Authors. This work is published and licensed by Atlanta Publishing House LLC, Atlanta, GA, USA. The full terms of this license are available at https://www.eurjchem.com/index.php/eurjchem/terms and incorporate the Creative Commons Attribution-Non Commercial (CC BY NC) (International, v4.0) License (http://creativecommons.org/licenses/by-nc/4.0). By accessing the work, you hereby accept the Terms. This is an open access article distributed under the terms and conditions of the CC BY NC License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited without any further permission from Atlanta Publishing House LLC (European Journal of Chemistry). No use, distribution, or reproduction is permitted which does not comply with these terms. Permissions for commercial use of this work beyond the scope of the License (https://www.eurjchem.com/index.php/eurjchem/terms) are administered by Atlanta Publishing House LLC (European Journal of Chemistry).





